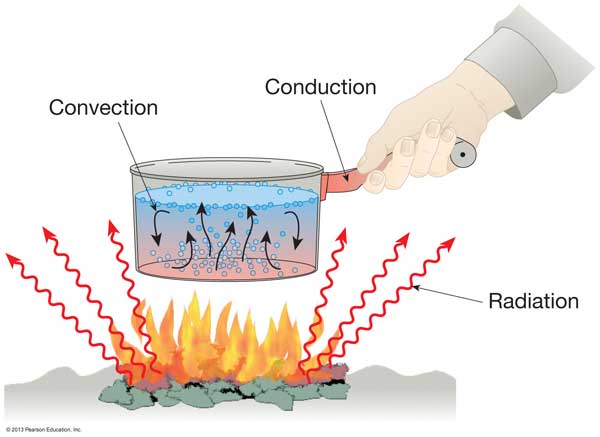
Modes of Heat transfer
- Heat is defined as the energy transfer without transfer of mass across the boundary of the system due to the temperature difference between the system and surrounding.
- The Process of heat flow from a higher temperature body to lower temperature body is heat transfer.
Heat transfer can be defined as transmission of energy from one region to another as a result of temperature difference or temperature gradient. - Heat transfer supplements the law of thermodynamics by providing additional experimental rules to establish energy transfer rates.
There are three different mechanism (modes of heat transfer) by which energy transfer is effected:
Conduction Heat Transfer
The heat is transferred from one molecule to the molecule situated next which is in contact; such a transfer of heat is called conduction.
The molecules do not move during heat transmission, so this mode of heat transfer can be take place in continues and rigid medium only. So we say that conduction take place in solid medium.
Convection Heat Transfer
The heat transfer due to the molecules actually moves from one point to another for transferring heat. Hence in this mode, the medium in which heat transfer takes place cannot be rigid but it should be continues. The heat transfer takes place in fluid.
In water tube boiler, water is flowing through the tube the hot gasses flow over the tube, heat is transferred from hot gas to metal tube and this heat is transferred to cold water flowing through the tube.
Radiation Heat Transfer
There is no need for any medium for transfer of heat can takes place in space also. The energy to start with will be converted into electromagnetic waves and these waves travel to any point through wave motion at very high speed, that velocity of light. The quantity of heat radiated depends upon absolute temperature of a body.
e.g. the heat transfer is the energy transfer from the sun to the earth.