Advantages and disadvantages of cold lime soda process
Category: Electronics Articles
8 Oct 14
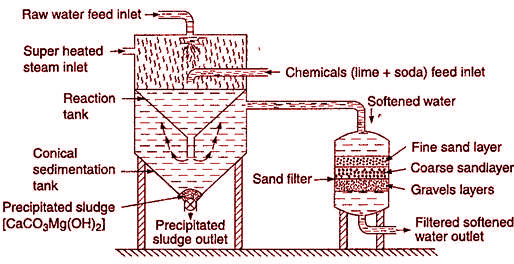
Cold lime soda process, when hard water is treated with calculated amount of lime [Ca(OH)2] and soda ash (Na2CO3) at room temperature 25ºC, the soluble Ca and Mg salt present in hard water are chemically converted into ppt. of calcium carbonate (CaCO3) and magnesium hydroxide [Mg(OH)2]. These ppts are removed by filtration. Thus soft water is obtained.
Advantages of cold lime soda process
- It can be carried out by four ways. So as per necessary, its required type can be used.
- Soft water obtained by this process has residual hardness about 50-60 ppm.
Disadvantages of cold lime soda process
- It is slow process because reactions during water softening takes place in very dilute solutions and room temp.
- It does not give soft water having residual hardness less than 50-60 ppm.
- It requires coagulant for setting particles of ppt. formed during reaction of water softening.
- Softening capacity of this process is less.
- Soft water obtained by this process consists of dissolved gases.